|
In addition, there were more-common side effects. Another suffered a stroke due to spending a long time in the operating theatre, and died. One patient developed infection at the surgery site, which caused multiple organ failure or sepsis, killing them. It is only recently I’ve started processing what I went through. Mrs Iacobucci had six surgeries in total before she gave up and demanded the device be removed. It just kept opening up and the leads would just pop through my skin.” A month after that surgery, I had another surgery to fix them again. “I had surgery to fix the leads, and then they became displaced again. But then, after a fall, she felt the electrodes come out of her spine. Lauren Iacobucci did receive good relief from her stimulator at first. And none of these people had received an opinion from a spinal surgeon.” That’s a pretty substantial investment to just throw in the bin. “In the last year, I have operated on three people … and taken out a stimulator and thrown it in the bin. “I think there are a moderate number of stimulators being put in inappropriately,” he said. Neurosurgery 1999 44:118-126.Michael Johnson, president of the Spine Society of Australia between 20, told The Age and Herald he was calling for the development of a spinal stimulation registry in Australia to monitor how well the devices were actually working, and how much harm they were causing. Magnetic resonance imaging with implanted neurostimulators: An in vitro and in vivo study. Tronnier VM, Staubert A, Hähnel S, Sarem-Alsani A. (testing of the Freedom-4 externally powered SCS). Evaluation of magnetic resonance imaging issues for a wirelessly powered lead used for epidural, spinal cord stimulation. MRI conditionality in patients with spinal cord stimulation devices. Feasibility of cerebral magnetic resonance imaging in patients with externalized spinal cord stimulator. (It may be possible to perform MRI in selected patients with "MR Unsafe" legacy SCSs if care is taken). 
Magnetic resonance imaging in patients with spinal neurostimulation systems. After the exam, MRI-mode should be turned off and settings returned to pre-MRI values. ĭe Andres J, Valía JC, Cerda‐Olmedo G et al. Visual and auditory monitoring should be continuously performed during the scan. The patient must be in a cognitive state able to provide immediate feedback concerning any problems during the examination. For several models there are restrictions on the use of transmit body coils and power deposition limits dependent on the location of magnet isocenter. Limits for maximum spatial gradient, gradient slew rate, total imaging time, and RF power dissipation measured by average SAR or maximum B 1+RMS must be followed explicitly. These conditions will vary significantly between manufacturers and SCS models. All current MR-conditional SCS devices are restricted to ¹H imaging in horizontal bore cylindrical magnets operating at 1.5 or 3.0T. Only the supine or prone patient positions are allowed. 
The IPG should be turned off or placed in "MRI Mode" if available. The IPG battery should be fully charged and lead impedances should measure within expected values. Pre-MRI evaluation/interrogation of the DBS system.Insure that the wires are connected to the IPG without breaks and there are no abandoned leads or electrodes. Verify (by x-ray if necessary) that the IPG has been implanted subcutaneously in the pelvis/flank. This can be done by reviewing operative notes from the medical record or the patient's information card. 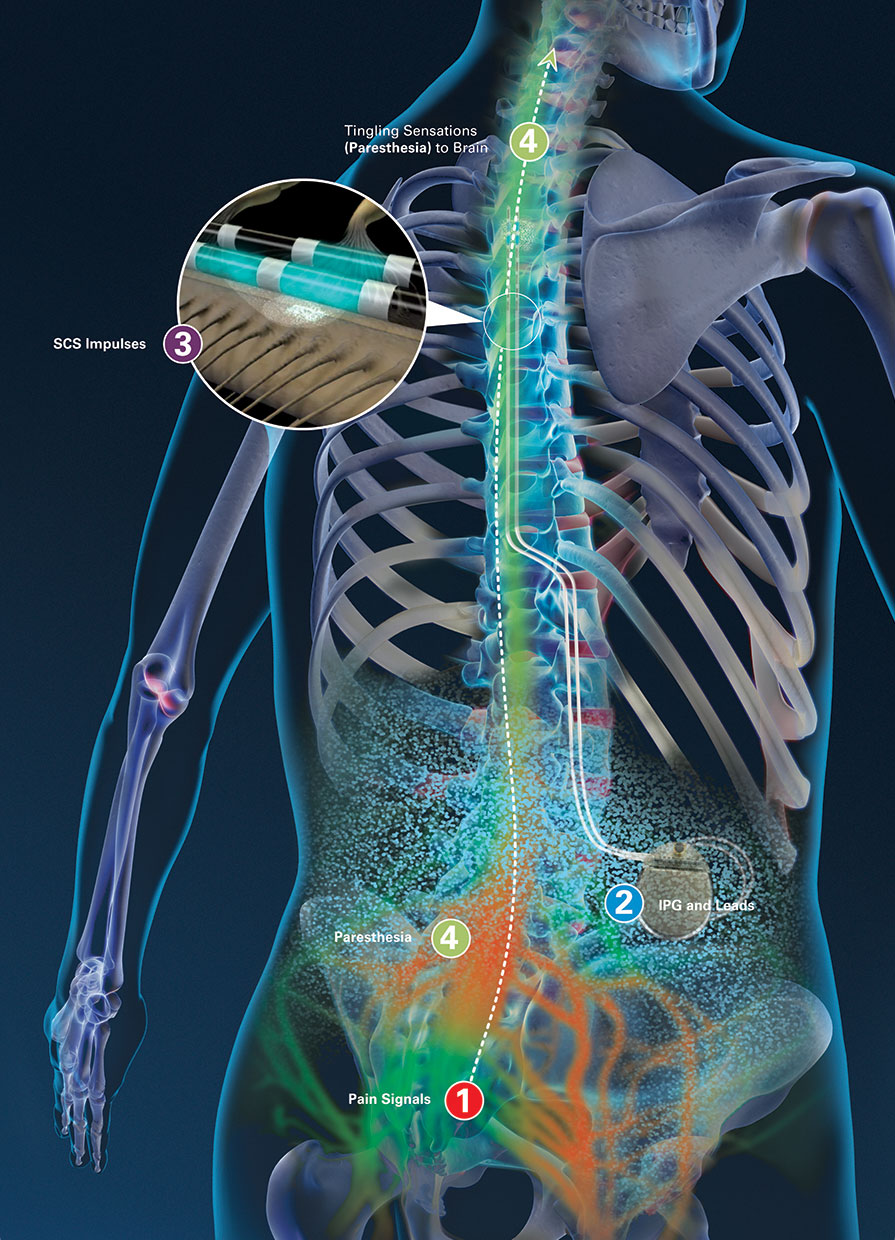
The exact model/serial numbers from the various components of the SCS system, including the IPG, leads, and extensions (if present) must be established with certainty.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
